Essential Medicines Initiative
Advancing essential medicines for critical care scenarios where current options are limited, compromised, or don't yet exist.
Glycan Engineering| CellFlow
Structurally defined glycans with enzymatic precision at synthetic speed.
Our Focus in Essential Medicines

Acute Care
Hospital & critical care settings
- Inpatient prophylaxis & treatment
- ICU, surgical, & emergency care
- Interventional & procedural support

Outpatient
Community & chronic care
- Post-discharge continuation therapy
- Chronic & episodic treatment
- Home-administered regimens

Preparedness
Strategic stockpile & field deployment
- Strategic national stockpile
- Forward & field deployment
- Mass casualty surgical triage

Acute Care
Hospital & critical care settings
- Inpatient prophylaxis & treatment
- ICU, surgical, & emergency care
- Interventional & procedural support

Outpatient
Community & chronic care
- Post-discharge continuation therapy
- Chronic & episodic treatment
- Home-administered regimens

Preparedness
Strategic stockpile & field deployment
- Strategic national stockpile
- Forward & field deployment
- Mass casualty surgical triage
Our Programs
Bioxaparin™ — Pioneering synthetic heparin
The world's most prescribed anticoagulant depends on a single animal-derived supply chain.
We engineered the synthetic alternative — improving supply security and patient safety.
patients treated annually
in the U.S.
API production in China
single-source dependency
pigs required per year
global supply chain
An essential medicine,
Our biosynthetic process emulates heparin's natural pathway through four sequential in vitro enzymatic steps — delivering a molecule structurally and functionally equivalent to Lovenox®.
The Insulin Story
% Insulin Volume Split by Type
Source: RethinkX, IMS Health, Washington Post, Novo Nordisk
Discovery & First Use
Insulin was discovered in the 1920s and produced from cattle pancreases for decades.
Recombinant Production
In 1982, the FDA approved Genentech’s synthetic human insulin — made from recombinant DNA, not animal tissue.
The Parallel
Within a decade, recombinant insulin became standard of care globally. Opening up the path to improved analog versions.
Tissue Repair & Regeneration
Heparin-Mimetic for prevention of acute organ/tissue damage
Acute organ damage destroys the endothelial and epithelial barriers that protect every organ.
Once compromised, fluid leaks, inflammation cascades, and tissue dies. From ARDS to sepsis to radiation injury — there are no approved therapies that directly restore these barriers.
A non-anticoagulant heparan sulfate mimetic engineered to restore barrier function across organs.
Stabilizes growth factors (FGF, VEGF)—accelerates repair
Preserves glycocalyx barrier—protects epithelium and endothelium
Neutralizes DAMPs & histones—dampens inflammation
Inhibits heparanase—prevents ECM breakdown & fibrosis
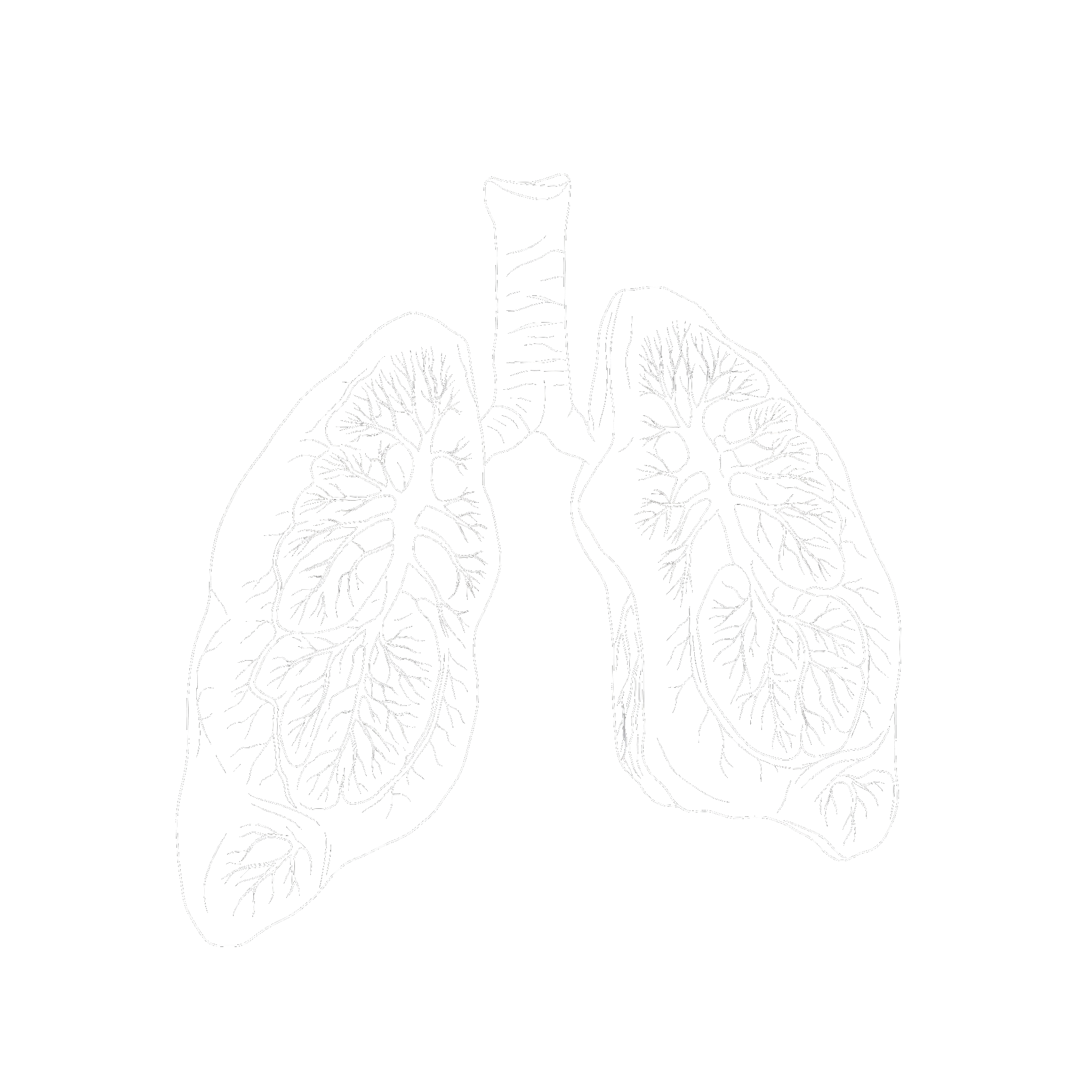
Acute Respiratory Distress Syndrome
ARDS destroys the alveolar-capillary barrier, causing fluid to flood the lungs. BIOX-2 stabilizes the endothelial glycocalyx to restore barrier integrity and protect lung tissue.
VOD/SOS Prophylaxis
Veno-occlusive disease damages the hepatic sinusoidal lining after bone marrow conditioning. BIOX-2 protects the liver endothelium during transplant conditioning to prevent barrier breakdown.
Sepsis & Systemic Inflammation
Sepsis degrades the glycocalyx systemically, causing vascular leak and cytokine-driven injury across organs. BIOX-2 neutralizes histones and DAMPs to dampen the inflammatory cascade.
Radiation-Induced GI Damage
GI mucositis after radiation or nuclear exposure destroys the gut lining. BIOX-2 preserves epithelial tight junctions and accelerates mucosal repair of the intestinal barrier.

Acute Respiratory Distress Syndrome
ARDS destroys the alveolar-capillary barrier, causing fluid to flood the lungs. BIOX-2 stabilizes the endothelial glycocalyx to restore barrier integrity and protect lung tissue.

VOD/SOS Prophylaxis
Veno-occlusive disease damages the hepatic sinusoidal lining after bone marrow conditioning. BIOX-2 protects the liver endothelium during transplant conditioning to prevent barrier breakdown.
Sepsis & Systemic Inflammation
Sepsis degrades the glycocalyx systemically, causing vascular leak and cytokine-driven injury across organs. BIOX-2 neutralizes histones and DAMPs to dampen the inflammatory cascade.
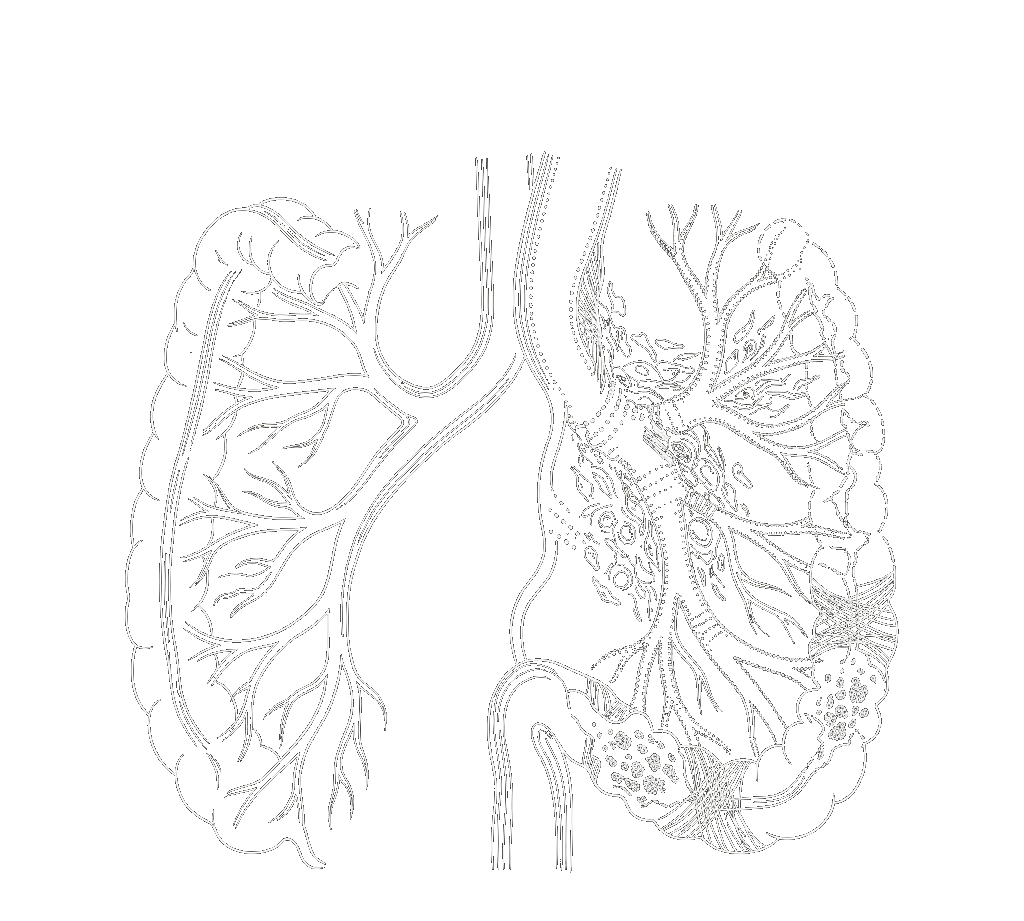
Radiation-Induced GI Damage
GI mucositis after radiation or nuclear exposure destroys the gut lining. BIOX-2 preserves epithelial tight junctions and accelerates mucosal repair of the intestinal barrier.
CellFlow Scale-Up

CellFlow Synthesis
U.S. Expansion with Partners
Distributed, scalable manufacturing — from factory floor to forward deployment.
- High-purity, modular scale-up
- Unprecedented biomolecule design space
- Precise control of branching, sulfation, and epimerization
Other Approaches
Limited design space, harsh reagents, expensive
Inconsistent structures, supply risk
Imprecise glycan control, low yields